What is a Battery?
A battery is an electrochemical device that can store energy in the form of chemical energy. It translates to electric energy when the battery is connected in a circuit due to the flow of electrons because of the specific placement of chemicals. It was invented by Alessandro Volta, whereas Gaston Plante invented the rechargeable battery.
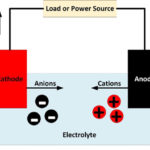
The battery consists of three elements: the negative side, the positive side, and electrolyte (the chemical which reacts with both sides), as shown in the image below. The electrolyte is used as an electron transportation medium between the anode and cathode.
It works due to electrochemical reactions called oxidation and reduction. In this reaction, electrons flow from one side to another side when the external circuit is connected to the anode and cathode.
The battery’s chemical composition can vary for different applications, specifications, sizes, etc., which are explained below in types of batteries.
Battery applications
The battery is used in applications where energy is required to be stored for future purposes. Portable, emergency, and low-power devices generally use batteries. A portable device, such as a mobile laptop, has a battery to use anywhere you want. An emergency device like an inverter, torch, etc., is used when there is no electricity. Low power devices like watches, oximeter, etc., can run for a long time after replacing the battery. The mains supply is not suitable for all situations.
The requirement of a battery depends upon various conditions like how much power is needed or what device portability is. But what about the wall watch? Why is this not connected to a socket?
The wall watch consumes very little power. A 1.2v battery can make it run almost for two years, but this is not the main reason. The watch should be powered up every instant to get the correct time; this can be done by battery. A single hindrance in power will cause a delay in time. That’s why it is designed to work with less power thereby allowing the watch to run for a longer period and making the battery an efficient way to supply power constantly.
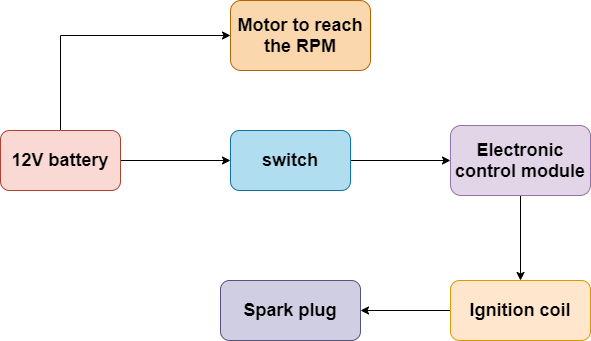
Let’s take another example. Generally, a vehicle works on petrol. In a self-start vehicle, the initial ignition of the engine is done by an ignition coil and a motor. The motor is used to reach specific rpm of the engine, and the ignition coil is used as a source of ignition. This vehicle ignition coil draws about four Amps. This current can vary among different manufacturers, and there is lots of space in the vehicle. That’s why to fill the higher current requirement lead-acid battery is perfect for it.
From the above example, we can say that the use of batteries depends upon condition and application.
Types of Batteries
Based on functionality, there are two types of batteries available in the market.
- Primary Batteries.
- Secondary Batteries.
Primary Batteries
The batteries made for one-time use only and unable to recharge, are called primary batteries. This type of battery is thrown away after use. It is also known as non-rechargeable batteries. It’s a very simple and convenient source of power for portable devices like a watch, camera, torch, etc. The battery comes in a standard size, as given below.
These batteries are cheap, small, lightweight, and there is no or low maintenance required.
Some common primary batteries
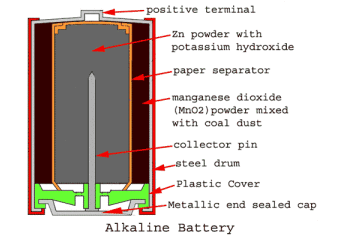
- Alkaline battery
The alkaline battery mainly consists of zinc and manganese dioxide as electrodes. The alkaline electrolyte is used as either potassium or sodium hydroxide. As you can see in the image below, the outer casing is a steel drum, and there is a cap on a drum which is a positive terminal. Inside this drum, the fine-grained manganese dioxide (MnO2) power mixed with coal dust is molted, as shown in the image. This molted mixture is part of the cathode in an alkaline battery. There is another powder filled inside the cathode powder, which is Zinc powder with potassium hydroxide. The Zinc (Zn) powder is part of the anode in an alkaline battery. Both powders are separated by a paper separator. The paper separator is soaked with potassium hydroxide, an electrolyte between the cathode (MnO2) and anode (Zn). The metallic brass pin is inserted along with the center axis of the alkaline battery, which is a negative collector pin. The pin is in touch with the metallic end. There is a plastic cover, which separates the metallic end and the steel drum. The metallic end is the negative terminal of an alkaline battery.
This battery is used where low voltage is required. One single cell can provide 1.5V. This is very cheap, so it can reduce the cost of the product. Every clock which hangs on the wall or remotes that control your TV and AC works on these alkaline batteries.
- Button cell battery
As you can see, the button cell is in the form of a button leading the body to be the cathode, and the anode is insulated at the top of the battery. The body is made of nickel-platted stainless steel – a positive terminal of the coin cell. At the top of the CAN, you can see a negative terminal cap. Both the CAN and the top cap are separated by a gasket made of insulator material. Inside the battery, there are two materials: Lithium metal and manganese dioxide, separated by a separator. The electrolyte used in the battery is lithium salt in an organic solvent.
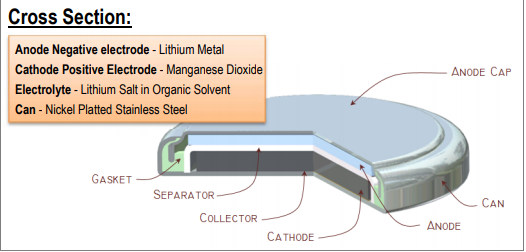
Button cell (Source)
Button or coin cells can be seen in watches in different sizes. This also comes in the alkaline batteries category because it comprises three substances- lithium as anode and manganese dioxide as a cathode, and alkaline as an electrolyte. These batteries are used to power small devices like watches, pocket calculator RAM, etc.
Secondary Batteries
The battery which is made for reusable purposes by recharging are called secondary batteries. They are also called rechargeable batteries. They have the same electrochemical reaction as alkaline batteries, but the electrochemical reaction can be reversed. This type of battery is used for portable devices like mobile phones, laptops, electric vehicles, etc. Also, a rechargeable battery is used with an inverter which stores power to supply our household devices.
Some common secondary batteries
- Lead-Acid batteries
The lead-acid battery container is made up of hard rubber of a bituminous compound. The container obtains dilute sulfuric acid, which is an electrolyte. The lead plates made of grid form are dipped in the electrolyte. The positive plate of the lead-acid battery is made of lead peroxide(PbO2). This is a dark brown hard, and brittle substance. The negative plate is made of pure lead in soft sponge conditions. A separator separates both electrodes. This separator can be made of cellulose, polyvinyl chloride, organic rubber, and polyolefins. The positive and negative are connected on the top of the battery, which is the outer positive and negative terminal to connect the load or device. There is a filter cap with a small hole in the center. The filter cap provides access for adding electrolytes, and the holes allow gases to be vented to the atmosphere.
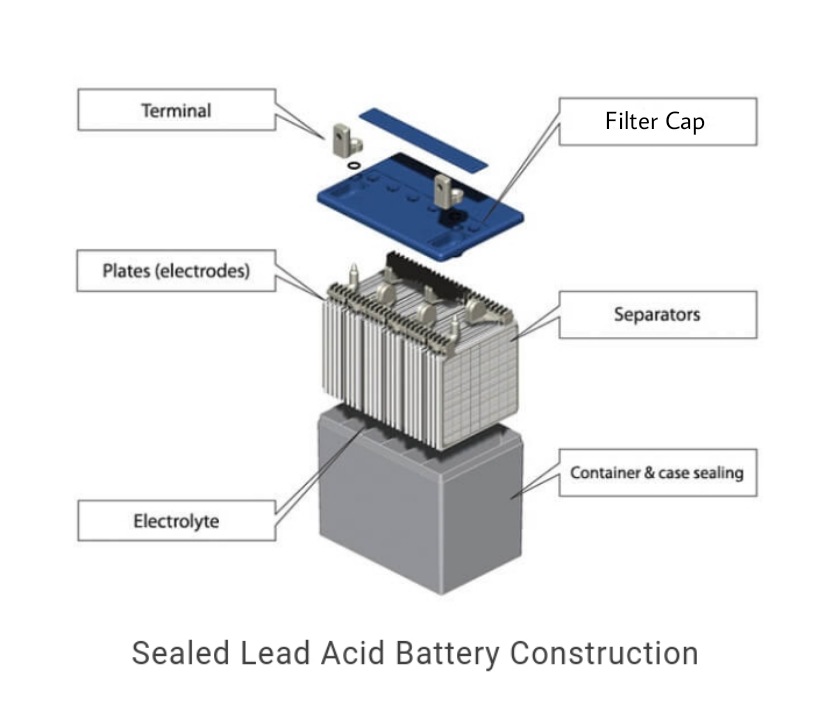
Lead Acid Battery (Source)
These batteries are low cost, reliable, larger, and are heavily weighted. It is mostly used in heavy-duty applications because it is not portable due to its weight and size. It is used in non-portable applications like solar-panel energy storage, vehicle ignition and lights, backup power, and load leveling in power generation and distribution.
- Nickel-Cadmium batteries
A Nickel-cadmium battery (Ni-Cd or NiCad battery) is made of nickel oxide hydroxide as cathode and metallic cadmium as an anode. Firstly, a layer of nickel oxide NiO2 is kept around the redox. This layer act as a cathode. Above this cathode layer, a separator of KOH or NaOH is made to provide OH ions. After this layer, the cadmium layer act as an anode of the ni-cd battery. Nickel layer act as a positive electrode and cadmium act as a negative electrode. The arrangement of the layer is rolled in a cylindrical shape in a case. The outer case is made of metal with a sealing plate and safety valve, which allow it to realize gasses out of the container. A cap on the top of the cell is insulated by a gasket, which acts as a positive of the ni-cd battery.
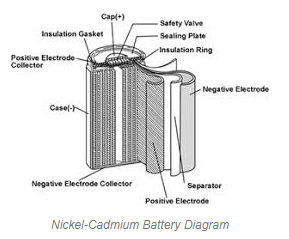
Nickel-Cadmium Battery. (Source)
These batteries are relatively less in cost, with toxic materials and a high self-discharge rate. It has a higher number of charging and discharging cycles. The energy density is higher than lead-acid batteries. It is smaller, lighter, and available in different sizes like alkaline batteries. It is generally used in low-cost devices like toys, solar light or cordless phones, etc.
- Nickel-Metal Hydride batteries
Nickel metal hydride battery (NiMH or Ni-MH) is made of Nickel oxide hydroxide as cathode and hydrogen-absorbing alloy as an Anode. The construction of the Ni-MH battery is the same as the Ni-cd battery. The Nickel oxide hydroxide layer and hydrogen-absorbing alloy are rolled with the separator of KOH or NaOH. The outer metal case Act as a negative terminal is connected with hydrogen-absorbing alloy. The cap on the top of the cell acts as a positive terminal and is connected with Nickel oxide hydroxide. An insulating seal ring or gasket separates both negative and positive terminals.
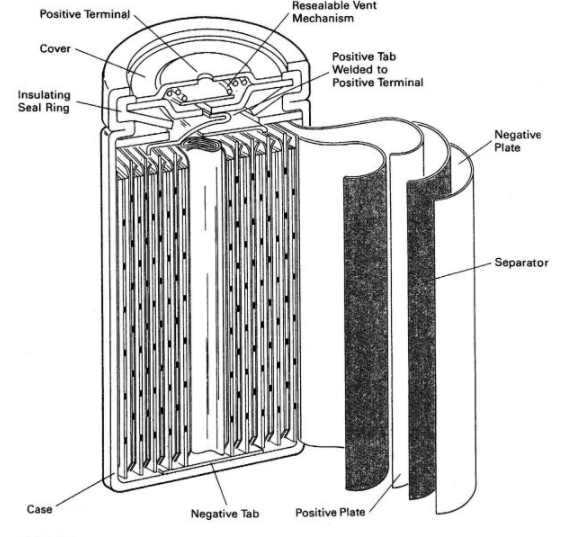
Nickel-Metal Hydride Battery. (Source)
Compared to Ni-Cd, these are more efficient with higher energy density, less toxic, and lower self-discharge rate. It is relatively expensive when compared to Ni-Cd. It has resistance to over-charging and over-discharging. It isn’t very easy to charge, and some manufacturers provide their specific chargers.
- Lithium-ion batteries
Lithium-ion batteries have anode made of graphite and cathode made of lithium metal oxide. The lithium salt as an organic solvent is used as an electrolyte. When the battery is connected to the circuit or load, lithium-ion migrates from the negative electrode to the positive electrode.
In the image below, the construction of the li-ion battery is similar to the Ni-Cd and Ni-NH batteries, apart from materials. The lithium metal oxide is coated on aluminum foil which is the positive electrode. The graphite is coated on copper foil which is the negative electrode. Both foils are rolled in a cylindrical shape with a separator between them. The spectator is soaked with electrolyte material which generally is lithium salt as an organic solvent. The outer metal casing is negative, and the top cap is the positive terminal. Both are separated by a gasket, which is made of insulating material.
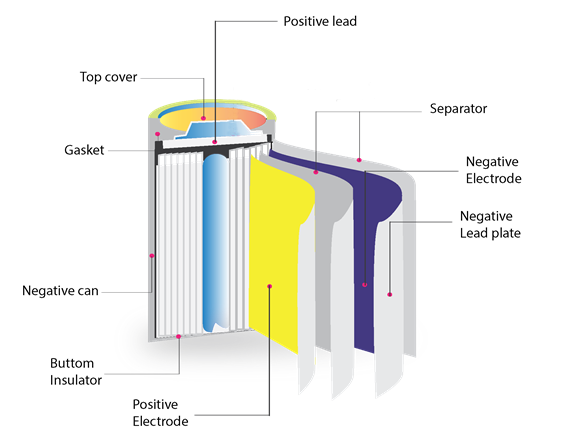
Lithium-Ion Battery. (Source)
Lithium-ion batteries are used in mobiles, laptops, and many portable devices. It is also used in the military and aerospace due to its lightweight nature. It has a higher energy density and low self-discharge compared to other types of batteries. It is also available in various sizes. Its single-cell voltage is higher. These have a significant risk of explosion when it is short-circuited or externally damaged.



Tell Us What You Think!