Separators in Lithium-ion (Li-ion) batteries literally separate the anode and cathode to prevent a short circuit. Modern separator technology also contributes to a cell’s thermal stability and safety. Separators impact several battery performance parameters, including cycle life, energy and power density, and safety. The separator increases internal cell resistance, and the separator takes up valuable space inside the Li-ion, making separator optimization an important part of Li-ion design. This FAQ briefly reviews separator operation and key performance metrics, reviews common separator materials for enhanced Li-ion safety, considers the possible use of functional separators that combine the operation of a separator and electrolyte, and closes with a look at UL 2591 Outline of Investigation for Battery Cell Separators.
Separators in Li-ions have to be electrochemically and chemically stable relative to the electrolyte and electrode materials since the separator is not part of the oxidation-reduction (redox) reactions that produce the flow of electricity (Figure 1). Separators operate under strongly oxidizing and reducing conditions and must be mechanically sound and able to withstand high stresses during battery assembly and operation.
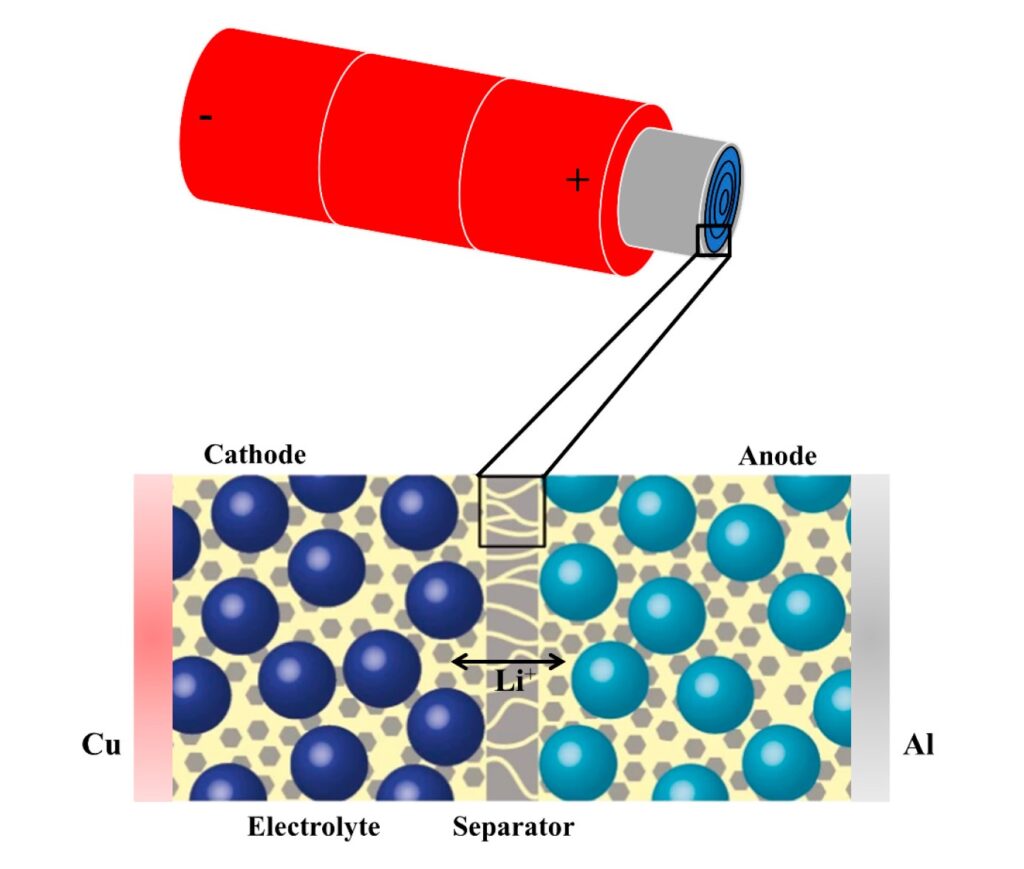
Figure 1: Schematic image of a separator in cylindrical Li-ion battery cell and a zoomed-in cross-section of the layered structure. (Image: MDPI molecules)
Separator materials include: Nonwovens consist of a manufactured sheet, web, or mat of directionally or randomly oriented fibers. Supported liquid membranes consist of a solid and liquid phase contained within a microporous separator. Some polymer electrolytes form complexes with alkali metal salts, which produce ionic conductors that serve as solid electrolytes. Solid ion conductors can serve as both separator and electrolyte. Regardless of the material used, battery separators are complex structures that must satisfy a wide range of sometimes conflicting performance requirements (Figure 2).
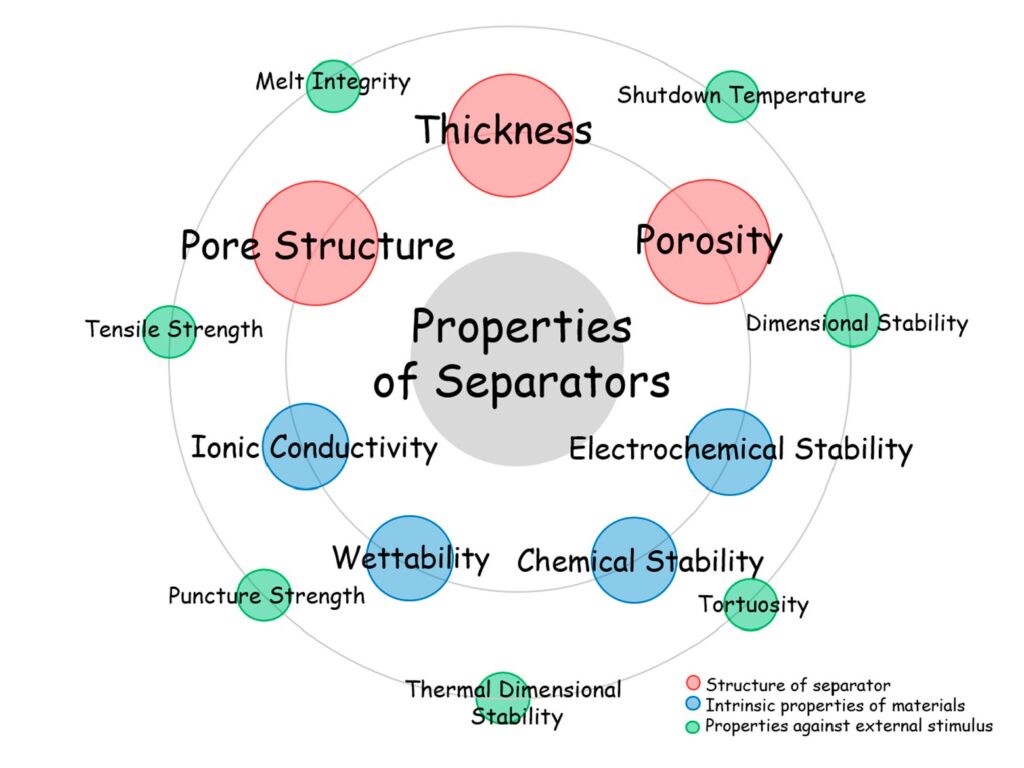 Figure 2: Important performance properties of separators. (Image: MDPI materials)
Figure 2: Important performance properties of separators. (Image: MDPI materials)
Chemical Stability: The separator material must be chemically stable against the electrolyte and electrode materials under the strongly reactive environments when the battery is fully charged.
Thickness & Strength: The battery separator should be thin enough to support the battery’s energy and power density and have sufficient tensile strength to prevent being stretched or damaged during the winding process. Separator thicknesses range from 25.4μm to 12μm, depending on the chemical system, without compromising the cell properties. The separator must not be punctured by particles or structures pressing on its surface to prevent electrical shorting.
Porosity & Pore Size: The typical porosity of a separator is 40 percent. If the porosity is larger, it can be difficult to close the pores during a battery shutdown event. The pores need to contain the electrolyte and allow ion movement between the electrodes. Pores should be uniformly distributed and have a tortuous structure, ensuring uniform current distribution throughout the separator while suppressing the growth of Li on the anode.
Thermal stability and shutdown: The separator thermally needs to be stable under normal operating temperatures and able to shut down at a temperature slightly lower than the temperature where thermal runaway occurs.
Wettability: The electrolyte needs to be compatible with the electrolyte ensuring complete wetting, and the electrolyte must be able to permanently wet the separator, preserving the cycle life.
Shutdown Separators
Shutdown separators are multilayer structures that can stop current flow in the battery if it gets too hot during the cycling process. At elevated temperatures, polymeric separators melt, and the pores close, stopping further ion transport in a mechanism known as separator shutdown (Figure 3). At extremely elevated temperatures, the integrity of the separator itself can be lost, resulting in a short circuit between the cathode and anode in a mechanism called separator breakdown. When separator breakdown occurs, it leads to thermal runaway.
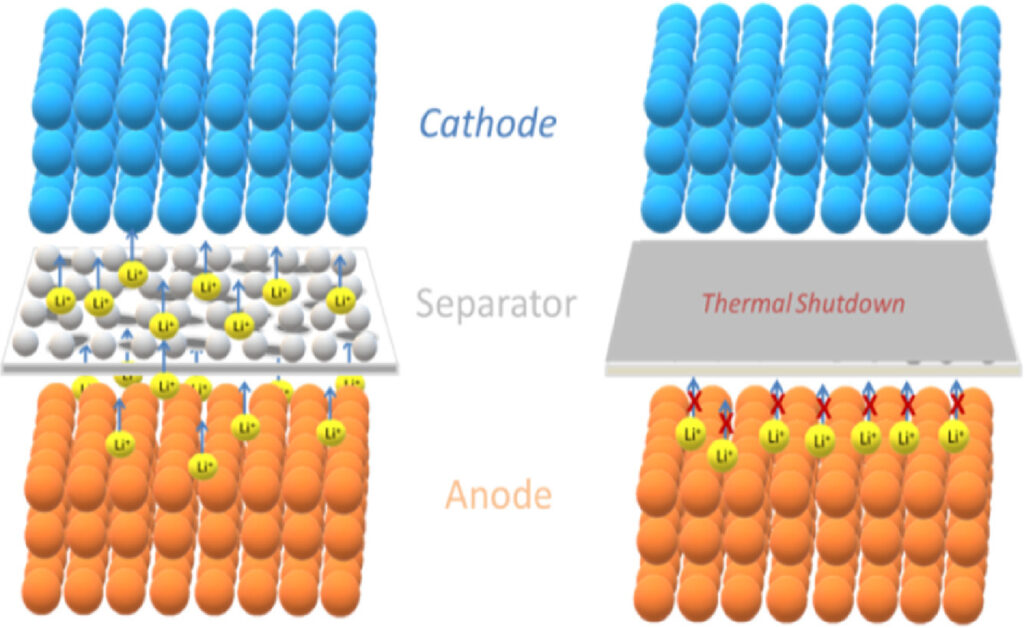
Figure 3 A polyethylene microsphere-coated separator can perform a rapid thermal shutdown at elevated temperatures to cut off the ionic transport and protect the battery from thermal runaway. (Image: Journal of Energy Chemistry)
Shutdown separators typically consist of polypropylene (PP)/polyethylene (PE) bilayer or PP/PE/PP trilayer structures. Shutdown separators take advantage of the difference in the melting point of PP (165°C) and PE (135°C). PE, with the lower melting point being the shutdown layer, and PP, with a higher melting point used to protect structural integrity and prevent separator breakdown. When an elevated temperature occurs, the PE layer melts, closing the pores in the separator and stopping current flow while the PP layer remains solid. If the temperature rises too rapidly and exceeds the melting point of PP, separator breakdown can still occur.
Ceramic-coated separators are made when ceramic particles such as alumina, zirconia, or silica are slurry-coated on polymer membranes. The thin ceramic coating reduces separator shrinkage upon thermal shutdown and improves battery safety. The addition of the coating also improves thermal and mechanical stability, and the high hydrophilicity and high surface area of the ceramic particles improves wettability. Still, it adds weight, volume, processing time, and cost to the separator. Ceramic-coated separators can also suffer from delamination from the polymer membrane leading to battery failure.
Functional separators and Li-metal batteries
So-called functional separators combine the capabilities of the separator and electrolyte. The development of functional separators will enable Li-metal batteries with capacities up to 7-times greater than today’s Li-ion batteries. Functional separators are expected to solve Li-metal battery problems related to dendritic growth, low Coulombic efficiency, high reactivity of Li-metal, and safety hazards.
Meta-organic frameworks (MOFs) are expected to be a key to developing functional separators. With a dense and highly-ordered framework of pores, MOFs have the largest know surface areas: One gram of MOF can have a surface area comparable to a FIFA soccer field, or 7 000m2/g of MOF material. The large surface area offers more space absorption of molecules and ions.
A functional separator consisting of a polypropylene separator coated on both sides with a MOF material could the enabler of Li-metal batteries. In the laboratory, a MOF with well-defined intrinsic nanochannels and negatively charged gap channels boh restricts the free migration of anions, contributing to a high Li+ transference number of 0.68. Meanwhile, the MOFs coating with a uniform porous structure promotes homogeneous lithium deposition. A highly-stable Li plating/stripping cycling has been achieved. However, numerous challenges remain to refine these materials and processes and scale them for high-volume production.
UL 2591 and battery cell separator safety
There are numerous safety and performance standards for Li-ions in specific applications such as automotive systems and consumer devices. But only one for battery cell separator safety: UL 2591, Outline of Investigation for Battery Cell Separators. To assess how different separator materials impact the safety of lithium-ion batteries, UL conducted a comprehensive assessment of lithium cobalt oxide (LiCoO₂) graphite pouch cells incorporating several types and thicknesses of battery separators including polypropylene, polyethylene, and ceramic-coated polyethylene with thicknesses from 16 micrometers (µm) to 7µm.
UL 2591 Ed. 3-2018 is the third edition of this standard and includes test procedures for battery cell separators for pore measurements (porosity, pore size, and distribution), wettability, and heat-of-combustion. Analytical tests to characterize battery cell separator material and the need to have a Quality Assurance program are also included. Revisions to Puncture Strength Test (inclusion of blunt-puncture test) and Dimensional Stability Test (inclusion of an alternate oven-method) were also made. Each sample cell underwent four testing evaluations to simulate common abuse conditions, including:
- Thermal ramp
- Overcharging
- Internal short circuit
- External short circuit
The separator materials used in the cell samples were also characterized for physical properties, including:
- Thickness
- Dimensional stability
- Melt temperature
- Melt rupture temperature
- Puncture resistance
- Shutdown function
Summary
Separators contribute to the safety and reliability of Li-ion batteries. When comparing various separator materials, there are numerous specifications, including chemical stability, mechanical strength, wettability, thermal performance and porosity, and pore size. Bilayer, trilayer, and ceramic-coated separators have been developed to prevent thermal runaway in Li-ion cells. Functional separators that use MOF-coated membranes to perform the dual functions of the electrolyte and separator are being developed to support the design of high-performance Li-metal batteries for high energy systems in electric vehicles and electric aircraft. While there are numerous performance standards for Li-ion batteries, UL 2591 is the only battery cell separator safety/performance standard.
References
A Review of Functional Separators for Lithium Metal Battery Applications, MDPI materials
A Review on Lithium-Ion Battery Separators towards Enhanced Safety Performances and Modelling Approaches, MDPI molecules
Elucidation of Separator Effect on Energy Density of Li-Ion Batteries, Journal of the Electrochemical Society
Safeguarding Lithium-Ion Battery Cell Separators, Underwriters Laboratories




I need some more information on battery materials and components