The electrolyte is often an underappreciated component in Lithium-ion (Li-ion) batteries. They simply provide an electrical path between the anode and cathode that supports current (actually, ion) flow. But electrolytes are a key to battery performance, and advances in electrolyte chemistries are expected to be an important development leading to high-performance, safe, and low-cost Li-ions for future generations of electric vehicles, grid-scale energy storage, and other large systems. Depending on the design of the battery, it can be a liquid or paste-like material. Solid electrolytes could enhance the energy density and safety of Li-ions, but they are not ready for commercialization.
This FAQ reviews the electrolytes in common use today and looks at advanced electrolytes under development, including solid-state and hybrid solid-state electrolytes.
Li-ion batteries consist of an anode and a cathode based on various redox chemical couples with an electrolyte and separator in between. The electrolyte conducts ions, not electrons, through the separator and between the anode and cathode (Figure 1). Electrolytes can take various forms, with dissolved salts being the most common form. The solvent is a key component of the electrolyte. Aqueous electrolytes have high conductivities (σ), typically σ = ∼1 Siemen/cm (S/cm), but have relatively small electrochemical stability windows of about 1.25V. Electrolytes based on inorganic solvents are less conductive with σ < 100 mS/cm but offer wider stability windows of 4V or more. In addition, organic solvents are more costly compared with aqueous solutions. Sometimes, a solid inorganic or polymer electrolyte is used, but with a much lower conductivity; σ <0.1mS/cm at room temperature.
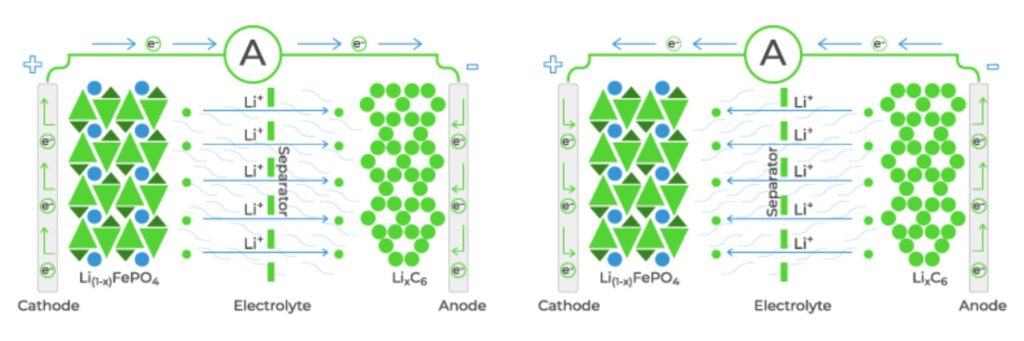
Figure 1: The electrolyte transports positively charged ions through the separator between the cathode and anode. (Image: dragonfly Energy)
Different primary (non-rechargeable) and secondary (rechargeable) battery chemistries rely on different electrolytes. Sulfuric acid serves as the electrolyte in most lead-acid batteries. Common alkaline primary cells use potassium hydroxide as the electrolyte. Salts such as lithium hexafluorophosphate (LiPF6) are commonly used as electrolytes in Li-ions.
Solid electrolytes for solid Li-ions
Solid-state electrolytes made with ceramics, such as lithium-metal oxides, promise higher energy densities and are not flammable, which will improve safety compared with liquid electrolytes. In addition to posing a risk of fires, liquid electrolytes can be highly reactive, leading to side reactions with the electrodes, resulting in lower battery capacity over time, called capacity fading. Batteries with solid-state electrolytes are not expected to experience capacity fading. Solid-state electrolytes are also expected to enable a high-capacity Li-metal anode that will significantly enhance the energy and power density and the cycle life of Li-ions. Development of solid-state electrolytes is complicated and requires a material with several performance characteristics:
#1 High ionic conductivity combined with high resistance to electrons
#2 High cation-transfer capability to support high power levels
#3 Wide electrochemical stability window for robust operation
#4 High thermal stability and high mechanical strength
#5 Easy and low-cost to manufacture
A commercially-viable material has yet to be developed: Solid-state electrolytes based on inorganic materials have high ionic conductivity and good mechanical strength. Polymer electrolytes are flexible, easier to process, and maintain good contact with the electrodes, reducing the interface resistance. But there are also challenges with each approach: Inorganic solid-state electrolytes can have poor contact with the electrodes (so-called grain boundary resistance), lack chemical and electrochemical stability, tend to be relatively expensive, and are brittle. Brittleness is a large challenge since inorganic solid-state electrolytes cannot compensate for the volume changes in the electrodes during charge and discharge. Polymer solid-state electrolytes can address many of the performance limitations of inorganic solid-state electrolytes. Still, they suffer from very low ionic conductivities, making them unable to support the charge and discharge rates needed for practical Li-ion designs.
So far, the choices for the best solid-state electrolyte have not been narrowed down; research efforts are underway on dozens of options for solid-state electrolytes, including sulfides, oxides, phosphates, polyether, polyester, polyurethane, and many more. At this time, polymers are easier to process than ceramics and have lower fabrication costs. Ceramics can be operated at higher temperatures and are more suitable for harsh environmental conditions.
Li-ions using solid-state electrolytes are expected to have very low self-discharge rates enabling multi-year energy storage with negligible losses. However, today’s materials have operating lifetimes of only about three years, making them impractical in almost all applications. Research continues because of the large payoff expected from the development of a viable solid-state electrolyte:
Energy density will be higher since solid-state batteries are over 80% thinner and have a higher decomposition voltage than today’s Li-ions. The high energy density would enable longer driving distances for electric vehicles (EVs) and reduce the number of charging stations needed to support EV operations.
Since solid-state batteries do not have a liquid electrolyte that gets heated during fast charging so faster charging is possible. Fast charging, combined with higher energy density, is expected to improve the marketability of future generations of EVs.
Solid-state electrolytes enable safe operation. Today’s liquid electrolytes are flammable and require numerous safeguards to ensure reliable operation. Solid-state batteries are inherently safe with very low risk of fire, and they can be operated at higher temperatures compared with Li-ions based on liquid electrolytes.
Lower cost will be possible with solid-state batteries. Liquid-based Li-ions cost about $220/kWh. While that is expected to continue declining, the rate of cost reductions is limited by the reliance on the use of scarce materials such as cobalt. Solid-state batteries are not expected to use scarce or rare materials. Using a solid-state electrolyte will eliminate the separator component, further simplifying battery designs, driving down the cost of Li-ions, and making them more attractive for use in EVs and large-scale energy storage systems.
Solid choices
One solid-state electrolyte that shows promise is made with lithium, scandium, indium, and chlorine (Figure 2). It has high ionic conductivity and low electron conductivity. It has a high electrochemical stability window that can support the development of a solid-state Li-ion that can operate without significant capacity loss for over a hundred cycles at high voltage (above 4 V) and thousands of cycles at lower voltage. This electrolyte’s chloride characteristics enable it to operate at over 4 V, making it suitable for use with the most common cathode materials used in current Li-ion designs.

Figure 2: Chlorine-based electrolytes may offer improved performance for solid-state Li-ions. (Image: Argonne National Laboratory)
Most previous solid-state electrolyte development focused on sulfides which are easier to work with but have a lower electrochemical stability window and degrade above about 2.5 V. To operate at higher voltages, sulfides require an insulating coating on the cathode that reduces the conductivity of the system. The substitution of a chloride chemistry for the sulfide eliminates that problem.
In addition to optimizing the solid-state electrolyte performance, optimization of the interface between the electrolyte and the anode and cathode layers is an important factor. One approach is to employ special coatings, which adds fabrication processes and cost.
An alternative method has been proposed that requires eliminating all carbon dioxide (CO2) present when the battery materials are sintered. Since the solid-state electrolyte, the anode, and the cathode are all made of various ceramics, the presence of even small amounts of CO2 can significantly reduce the performance of the interfaces between the electrolyte and the anode and cathode. Work continues in various aspects of solid-state electrolyte formulations and fabrication. Hybrid electrolytes may provide a near-term alternative.
Hybrid electrolytes
A recently-proposed way to address the performance limitations of inorganic and polymer solid-state electrolytes is to hybridize two or more elements. Development of a commercially-viable hybrid solid-state electrolyte (HSE) could overcome the disadvantages of both inorganic and polymer technologies while combining the strengths of each technology (Figure 3).
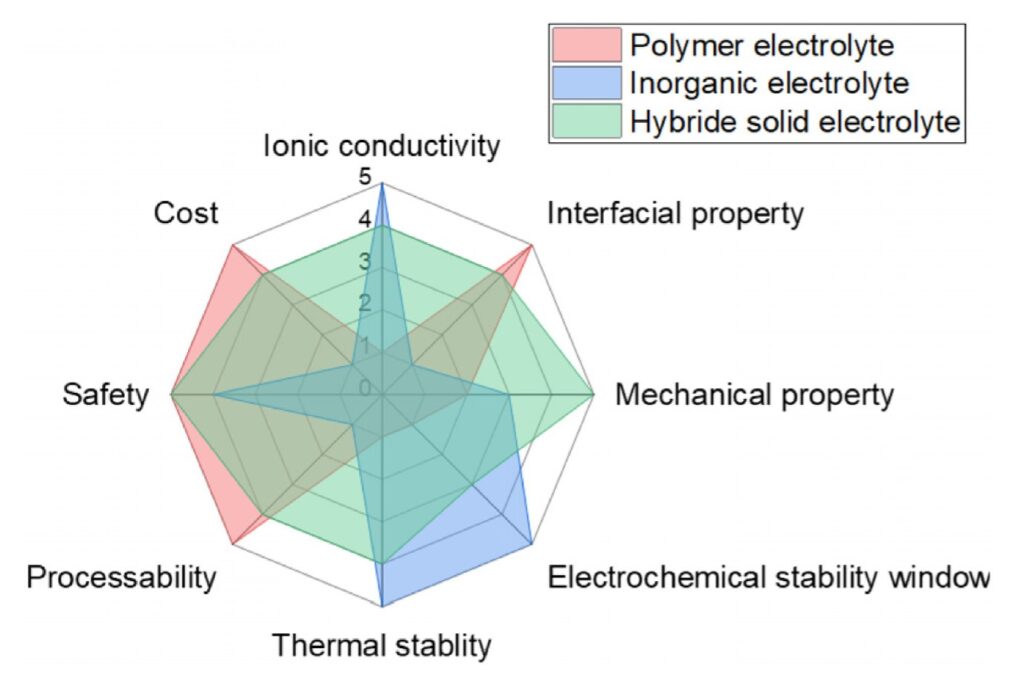
Figure 3: Performance comparison of polymer, inorganic and hybrid solid electrolytes. (Image: Frontiers in Energy Research)
As with all solid-state electrolytes, developing HSEs is challenging, and a commercial solution is still in the future. HSEs have better ionic conductivity than most polymer electrolytes, but it’s still too low. In addition, HSEs suffer from poor interfacial stability (reducing reliability), and high interfacial resistance (reducing charge/discharge rates), which greatly limit the performance of HSE-based Li-ions.
Summary
Li-ion batteries with various liquid electrolytes provide good combinations of performance and safety. But they are relatively costly. New types of electrolytes will be required to improve Li-ion performance, drive down costs, and increase safety. Numerous efforts are underway to develop solid-state electrolytes that promise Li-ions with higher energy densities, faster charging, safer operation, and lower cost. The emergence of HSE promises to accelerate the development of solid-state Li-ion batteries.
References
Avoiding CO2 Improves Thermal Stability at the Interface of Li7La3Zr2O12 Electrolyte with Layered Oxide Cathodes, Advanced Energy Materials
Battery Materials Design Essentials, ACS Publications
Electrolyte Design for Fast-Charging Li-Ion Batteries, Trends in Chemistry
Recent Developments and Challenges in Hybrid Solid Electrolytes for Lithium-Ion Batteries, Frontiers in Energy Research
Scientists discover new electrolyte for solid-state lithium-ion batteries, Argonne National Laboratory
What Is Battery Electrolyte and How Does It Work?, dragonfly Energy





Tell Us What You Think!