Researchers at Chung-Ang University used high-concentration electrolytes to improve safety and prevent cell swelling caused by lithium plating.
To achieve a greater adaption of electric vehicles, the automotive industry needs fast-charging batteries that don’t compromise energy density. Lithium plating for ultra-fast charging raises safety concerns due to high cell polarization, requiring careful attention.
During fast charging, lithium-ion batteries can face safety issues caused by polarization. Researchers at Chung-Ang University in Korea studied the impact of high-concentration electrolytes on battery interface kinetics and stability. The research demonstrates effectiveness in improving fast charging and preventing issues such as cell swelling, which can result from lithium plating. According to a press release, this study provides insights for future electrolytes that can enhance the extremely fast-charging abilities of Li-ion batteries.
Associate Professor Janghyuk Moon of Chung-Ang University led the study, published in Energy Storage Materials titled Boosting interfacial kinetics in extremely fast rechargeable Li-ion batteries with linear carbonate-based, LiPF6-concentrated electrolyte. The research highlights a strategy to mitigate polarization resulting from concentration-based effects. In the press release Dr. Moon noted “Our study aims to enhance the range and reduce charging times of electric vehicles by developing advanced battery technologies, specifically leveraging the commonly used LiPF6 and linear carbonates in commercial batteries. By improving the kinetics and stability of batteries under fast charging conditions, we hope to make a meaningful impact on the EV industry and ultimately on people’s daily lives.”
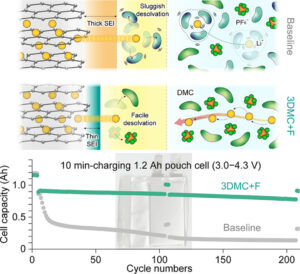 This study investigated how using linear carbonate-based electrolytes such as ones with concentrated LiPF6, can affect the desolvation process, which removes the solvent from Li-ion and its quick insertion into the graphite anode. The researchers used electrolytes with low activation energy, including dimethyl carbonate (C3H6O3), ethyl methyl carbonate (C4H8O3), and diethyl carbonate (C5H10O3) with a special focus on dimethyl carbonate. These electrolytes made it easier to overcome the desolvation barrier. Tests confirmed that these electrolytes enhance the fast-charging capabilities of Li-ion batteries, evidenced by a 1.2-Ah pouch cell retaining three times more capacity over 200 cycles. The process also prevented cell swelling caused by Li plating, a common issue with traditional electrolytes.
This study investigated how using linear carbonate-based electrolytes such as ones with concentrated LiPF6, can affect the desolvation process, which removes the solvent from Li-ion and its quick insertion into the graphite anode. The researchers used electrolytes with low activation energy, including dimethyl carbonate (C3H6O3), ethyl methyl carbonate (C4H8O3), and diethyl carbonate (C5H10O3) with a special focus on dimethyl carbonate. These electrolytes made it easier to overcome the desolvation barrier. Tests confirmed that these electrolytes enhance the fast-charging capabilities of Li-ion batteries, evidenced by a 1.2-Ah pouch cell retaining three times more capacity over 200 cycles. The process also prevented cell swelling caused by Li plating, a common issue with traditional electrolytes.
The study also utilized “molecular dynamics”release to theoretically simulate liquid electrolyte structures at varying concentrations, both high and low. These aimed to understand micro-environmental changes within the battery system, offering insights into how the electrolyte affects interface kinetics and battery performance. This combined approach of practical experimentation and computational analysis highlights the significance of this study in advancing battery technology for practical applications, particularly in electric vehicles.





Tell Us What You Think!