By JD DiGiacomandrea, Green Cubes Technology
Learn why Lithium-ion-phosphate batteries need the right battery-management system to maximize their useful life. It’s all about chemistry.
Lithium-ion (Li-ion) batteries provide high energy density, low weight, and long run times. Today, they’re in portable designs. Their popularity has spawned a few sub-chemistries that all use the principle of shifting lithium ions from anode to cathode. Different versions of these anodes and cathodes offer different performance characteristics. The prevailing nickel manganese cobalt (NMC) chemistry is beginning to be displaced by a “new” sub-chemistry, Lithium Iron Phosphate (LFP or LiFePO4).
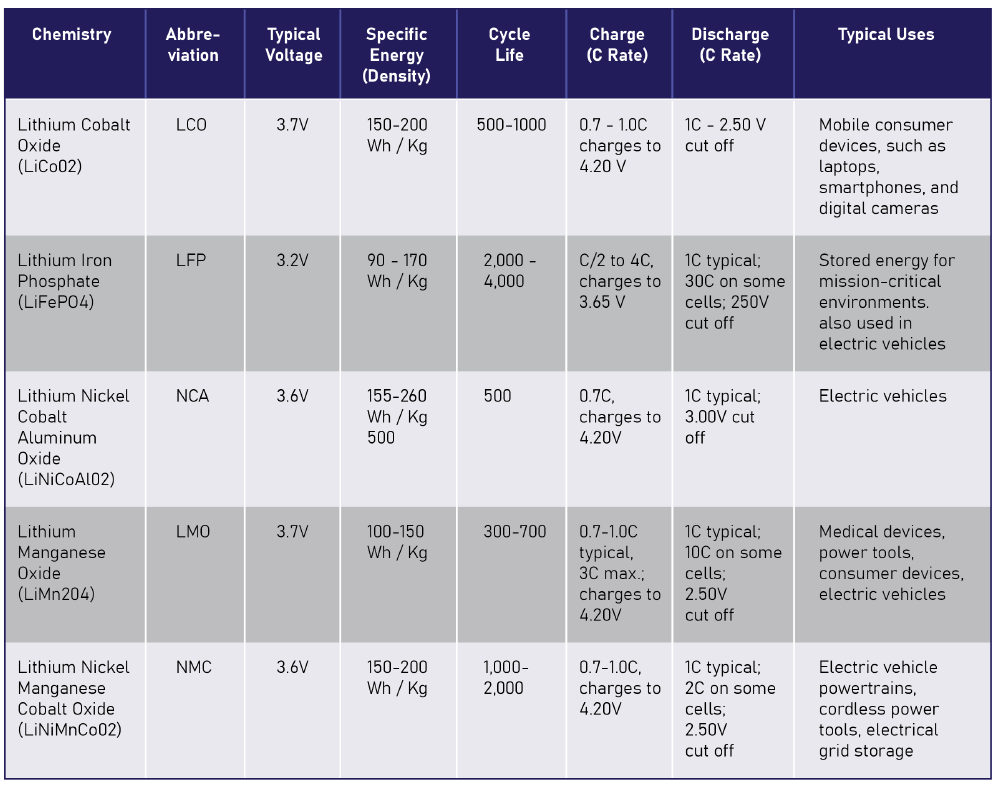
Table 1 compares the most common lithium-Ion chemistries. There are several factors that may sway you to choose LFP over NMC including technical, political, and performance. As a designer, you should note the specific differences between NMC and LFP and consider them in your battery design.
NMC chemistry has been the most popular lithium-based chemistry in automotive, consumer, and industrial markets for the better part of 20 years. In the past ten years alone, the adoption of NMC into EVs has severely impacted the NMC supply chains. Factories have produced cells at capacity; there are more factories being built in the next three years than ever before. Additionally, rare-earth elements are in high demand (with volatile pricing, swinging over 200% or more in 6 months. https://tradingeconomics.com/); particularly nickel and cobalt (the N and C in NMC, M is manganese). Due to the high demand, the prices have been very unstable — tripling in 2018 and again in 2022. for devastating landscapes, water pollution, contaminating crops, and loss of soil fertility while being subject to poor working conditions and abusive labor practices. For these reasons, designers look for alternative battery chemistries that don’t require cobalt and nickel.
LFP was developed by A.K. Padhi, K.S. Nanjundaswamy, and J.B. Goodenough at the University of Texas at Austin in 1996. John B. Goodenough developed the initial LCO lithium-ion chemistry in the 1970s and later went on to win the Nobel Prize for his role in the development of lithium batteries. Developed by using the natural mineral in the olivine family — LiFePO4 as the cathode and a graphite carbon on a metallic backing (current collector) as the anode — the LFP battery was developed and optimized for use in portable electronics over the next decade.
LFP versus NMC
LFP has a few distinct advantages over NMC. It is extremely temperature stable, meaning the chemistry doesn’t break down at high or low temperatures. The impedance is more stable than NMC over the standard temperature range of -20°C to 65°C. LFP is also much safer and resistant to thermal runaway than NMC because of its extremely strong oxygen bond in the compound. That bond prevents an oxidizer from being available during a redox reaction. If you were to conduct a test, you’ll see that LFP will merely heat, steam, and vent while NMC will usually ignite and throw showers of flame and sparks during a thermal runaway event. LFP also boasts 3000 to 10,000 cycles before the cell degrades to 80% of its original capacity. Finally, LFP is non-toxic and uses abundant inexpensive elements including iron and phosphorus.
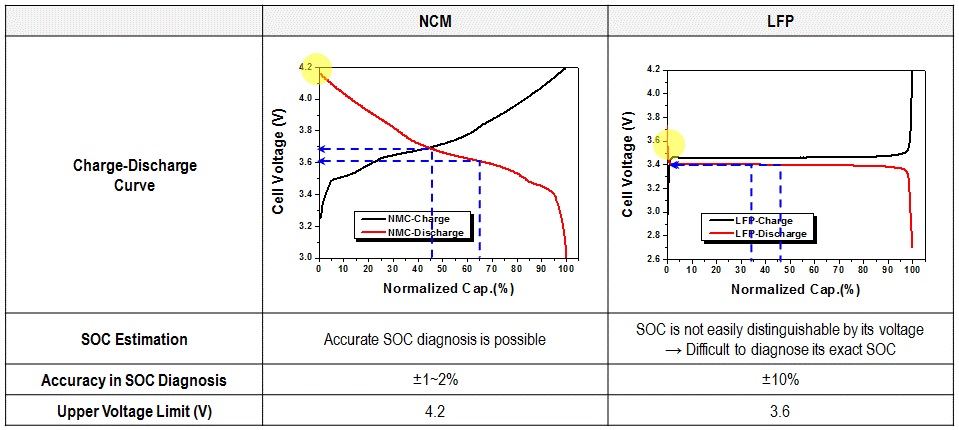
Though LFP has disadvantages that affect applications. You must consider these challenges in your designs because they can adversely affect the user’s experience. First, LFP has a very “flat” voltage curve, see Figure 1. The voltage range of a single LFP cell is 2.5 V to 3.65 V, but from 90% to 10% state-of-charge (SOC), the voltage is between 3.1 V and 3.3 V. This flat voltage curve prevents simple voltage to SOC relationships. Many off-the-shelf fuel-gauge circuits for lead acid, nickel metal hydride, and alkaline cells use a simple voltage measurement for SOC calculations. Unfortunately, this will not work on LFP cells. You should investigate either a coulomb-counting algorithm or an advanced algorithm such as the Texas Instruments Impedance Track algorithm.
Coulomb counting uses a current-monitoring device such as a shunt or hall-effect sensor to detect the current passed into or out of the battery. Combined with a simple voltage monitor, coulomb counting lets you estimate the SOC of an LFP cell fairly well. It’s important to consider that this algorithm is prone to drift over time due to the calibration of the current sensor and will require periodic corrections. You can then detect a fully charged condition (>3.6 V/cell) and fully discharged (<2.6 V/cell) condition periodically and calibrate the algorithm at those points. The disadvantage to this method is it may not consider capacity loss due to cold temperatures or aged cells. A more advanced algorithm such as the Impedance Track algorithm has the capability to combine coulomb counting and voltage corrections with temperature data and historic trends of capacity to continually adjust the learned capacity of the battery. These algorithms require you to dial in the parameters in the settings to their specific application, which is beyond the scope of this article.
LFP also has the advantage and disadvantage of extremely low internal impedances. The advantage comes because LFP cells can provide high discharge currents with little heat generation. They also operate at high charge rates in applications that require fast charging. Unfortunately, you must consider that during a short-circuit event, the battery can quickly deliver large currents. These large currents must be controlled by a battery-management system’s current control device. Typically, larger battery packs use a MOSFET or contactor.
The designer must ensure the MOSFET’s turn-off time (operating in the linear region) and that the drain current does not exceed the safe-operating region. This may require the use of a separate MOSFET driver circuit as typical drivers built into off-the-shelf battery-management system (BMS) chipsets are limited. Another common issue arises because this turn-off time may be very short; the dI/dT may become very large and cause ringing on the current conduction path, which may exceed the MOSFET’s voltage rating. The inductance of the cabling connecting the cells to the BMS will cause this problem. To alleviate this ringing and voltage rise, you can place an RCD circuit (Figure 2) near the MOSFETs to divert excess energy to the capacitor. This circuit simply requires a resistor, capacitor, and diode to dump excess energy and then slowly dissipate the energy in the resistor.

Figure 2. This RCD snubber circuit prevents high voltages from building across a MOSFET when the feed cable acts as an inductor.
LFP also has a higher self-discharge rate than other NMC chemistries. This self-discharge rate is defined as the loss of capacity internal to the battery over time. This capacity is lost through small discharge currents inside of the cell and is typically on the order of 2% to 10% capacity per month. The more controlled the manufacturing environment, the lower the self-discharge rate and the more consistent it can be between cells. If your cell manufacturer has a poorly controlled manufacturing process, the self-discharge rate can be nearly 10% per month on bad cells, and less on good cells, varying widely. This can cause imbalance issues in the battery once assembled, especially if the battery is put into storage before use.
To alleviate this issue, you should select a cell vendor with strong quality control policies and ensure your design has a robust cell-balancing function. Typical lithium-ion battery management systems all include some sort of cell balancing, with passive cell balancing being the most common. This is accomplished by using cell-voltage monitoring and turning on a bleed resistor when a cell voltage exceeds a predefined threshold of typically 3.4 V to 3.5 V. Thus, the balancing function only operates during a charge session, wasting little energy. The size of the passive resistor must cause the cells to discharge at a rate that balances the balancing speed and heat dissipation for resistor size and location. This is typically a 300 mA discharge per 100 Ah of cell capacity, though it should be selected based on the application.
Balancing act
Because this balancing is not operational during storage, often LFP batteries can become very imbalanced during long periods of storage; each cell will self-discharge at a slightly different rate. Here is where user education can be the best solution.
Having a warning that after long periods of storage, the battery must be charged and left on the charger for an extended period to “re-balance.” Doing so prevents users from removing a battery from long-term storage and finding the runtime extremely low compared to their expectations.
Finally, LFP as a chemistry requires some mechanical pressure to keep the anode and cathode in contact with the separator, prevent delamination, and optimize the cycle capabilities. The LFP chemistry swells slightly between charge and discharge. LFP cells are manufactured in numerous physical configurations. They can be manufactured in a cylindrical can, foil pouch, or a prismatic cell. A prismatic cell is usually contained in a rectangular-shaped aluminum or steel box (Figure 3), with terminals on the top for welding or screws. In cylindrical configurations, the round shape of the cell provides mechanical pressure to ensure the anode and cathode remain in contact with the separator.
In prismatic cells, there is a potential weak point where the largest surface of the cell is in the same plane of chemistry’s expansion and contraction. This requires some external pressure plates to apply pressure to the cell assembly and optimize the cycle count. If pressure isn’t placed on the cells, the cell’s capacity will rapidly degrade as the cell is cycled. Typical configurations of prismatic cells place all the cells together, and then pressure plates are placed on either end, with metal or plastic straps used to hold the entire assembly together.
As LFP cells are considered safer than NMC cells, you should remember that all lithium-ion batteries should be designed to prevent short circuits, charging after deep discharges, and overcharging. These are the primary causes of overheating and thermal runaway. Thorough testing of any BMS design should include deep discharging to below 2.0 V per cell and then attempted recharge. This is when copper that has dissolved into the electrolyte can be redeposited on the anode in an uneven way, causing the formation of dendrites that can puncture the separator and cause internal short circuits.
Short circuits cause high currents, which cause hot spots that can lead to thermal runaway. To prevent dendrite formation, all BMSs should limit the rate of charge after a deep discharge event to C/100 or lower until the cell voltage recovers to greater than 3.0 V or so. There also should be a time limit, to prevent the cell voltage from increasing above 3.0 V after X hours of charge at C/100. Where X is an appropriate number based on the capacity of your battery system and charge rate, for example, 30 minutes at C/100. At that point, the battery should be considered damaged, and the BMS should disable all future charge for safety reasons. BMS designs often don’t include this feature.
As you can see, LFP chemistry can be a great solution for our portable designs. It offers optimal performance for a lower cost and a significantly higher cycle life than NMC. There are some nuanced performance differences in LFP when compared to NMC that must be considered in BMS design. Most importantly, to design a safe, stable, and higher-performing lithium iron phosphate battery, you must test your BMS designs early and often, and pay special attention to these common issues. Every lithium-ion battery can be safe if the BMS is well-designed, the battery is well-manufactured, and the operator is well-trained.
About the author
JD DiGiacomandrea is the Product Marketing Engineer for Green Cubes Technologies. As a Lithium battery and energy storage industry veteran JD has over a decade of experience designing Lithium batteries and systems for the military, medical, and industrial markets. JD has had roles at major battery manufacturers including Electrical Engineer, Applications Engineer, and Field Sales Engineer, bridging the gap between Engineering and Sales. JD holds a Bachelor of Science in Electrical Engineering from Clarkson University in Potsdam, NY and a PE license from the State of New York.




Tell Us What You Think!