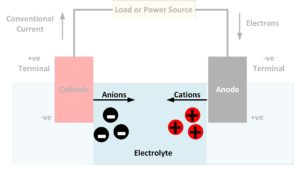
The electrolyte in a battery is the substance that allows electrical current to flow between the anode and the cathode. Electrolytes may be fluids or solids. Soluble salts, acids, and bases can generally act as electrolytes. While current flows through a metallic conductor in the form of lone electrons, within an electrolyte current flows in…
Battery pack protection keeps EVs on the road
A few best practices for handling transients and over-currents help make electronic drivetrains more reliable. James Colby • Littelfuse, Inc. Automotive market analysts predict that by 2025, high-end vehicles will contain more than $6,000 in electronic components. Most of these advanced electronics will be in the growing number of electric vehicles (EVs). To ensure robust, […]
What is an anode?
An anode is an electrode where oxidation reactions occur, involving atoms giving up electrons. The negatively charged free electrons then flow out of the negative terminal of the battery to produce an electrical current. It should be noted that conventionally, electricity is considered to flow in the opposite direction to the flow of electrons. Anions,…
Say goodbye to cross-country road trips
Leland Teschler • Executive editor Officials estimate it will cost $50 billion to field the chargers needed for the expected number of electric vehicles on U.S. roads by 2030. But it’s possible that spending every penny of this amount won’t prevent the extinction of the cross-country road trip. Here’s the logic behind that statement: It’s […]
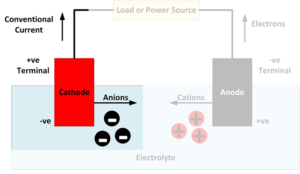
What is a cathode?
A cathode is an electrode where reduction reactions occur, in which atoms gain electrons. Negatively charged free electrons flow into the positive terminal of a battery as an electrical current. Because electrons are negatively charged, electricity is conventionally considered to flow in the opposite direction to the flow of electrons. Cations, positively charged atoms which…
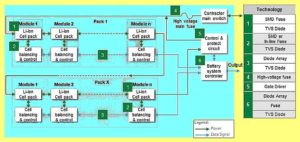
Understanding battery management systems
A battery-management system (BMS) is an electronic system or circuit that monitors the charging, discharging, temperature, and other factors influencing the state of a battery or battery pack. It’s used to monitor and maintain the health and capacity of a battery. Today’s BNS devices are advanced and will often provide pop-up notifications as you’ve likely…
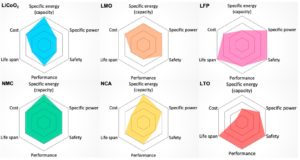
The difference between primary and secondary battery chemistries
There are several differences between primary and secondary batteries. The most obvious is that primary batteries are single-use devices while secondary batteries can be recharged and used many times, but that’s not the only difference. This FAQ starts with a general overview of the differences between primary and secondary batteries; it will then look at…
How to read battery discharge curves
Batteries are complex electrochemical and thermodynamic systems, and multiple factors impact battery performance. Of course, battery chemistry is at the top of the list. Still, factors such as charge and discharge rates, operating temperatures, storage conditions, physical construction details, and more come into play when understanding which battery best suits a specific application. To begin,…
The difference between lithium ion and lithium polymer batteries
A lithium-ion polymer (LiPo) battery (also known as Li-poly, lithium-poly, PLiON, and other names) is a rechargeable Li-ion battery with a polymer electrolyte in the liquid electrolyte used in conventional Li-ion batteries. There are a variety of LiPo chemistries available. All use a high conductivity gel polymer as the electrolyte. LiPos provide higher specific energies…
Introduction to batteries and their types
What is a Battery? A battery is an electrochemical device that can store energy in the form of chemical energy. It translates to electric energy when the battery is connected in a circuit due to the flow of electrons because of the specific placement of chemicals. It was invented by Alessandro Volta, whereas Gaston Plante…